Absorbable surgical sutures are a cornerstone of modern healthcare, playing a critical role in wound closure across a wide range of surgical procedures. Unlike non-absorbable sutures, which require removal, these sutures gradually break down in the body over time, eliminating the need for follow-up visits to take them out. This key feature not only improves patient comfort but also reduces the risk of complications associated with suture removal, such as infection or tissue damage. In recent decades, significant advancements in polymer science and manufacturing technology have led to the development of synthetic absorbable sutures that offer superior performance compared to their natural counterparts, such as catgut. This article explores the latest innovations in absorbable surgical sutures, their clinical advantages over traditional options, and the manufacturing processes that ensure their safety and efficacy.
What Are Absorbable Surgical Sutures?
Absorbable surgical sutures are medical devices designed to hold tissues together during the healing process and then degrade naturally within the body, eliminating the need for removal. They are typically made from either natural materials (e.g., catgut, derived from animal intestines) or synthetic polymers (e.g., polyglycolic acid, PGA; polyglactin 910, PGLA). The choice of suture depends on several factors, including the type of surgery, the location of the wound, the patient’s healing rate, and any potential allergies or sensitivities.
The primary function of absorbable sutures is to provide sufficient tensile strength to maintain wound closure until the tissue has healed enough to support itself. The rate at which the suture loses strength and degrades is carefully controlled to match the healing timeline of the tissue. For example, a suture used in skin closure may need to maintain strength for 7-10 days, while one used in internal tissues (like the abdomen) may need to hold strength for several weeks.
Key Types of Absorbable Surgical Sutures and Their Properties
Modern absorbable sutures come in a variety of types, each with unique properties tailored to specific clinical needs. Below is a detailed breakdown of the most common types, including those manufactured by leading producers like Suzhou Sunmed Co., Ltd.:

1. Polyglycolic Acid (PGA) Sutures
PGA is a synthetic polymer derived from glycolic acid, a naturally occurring substance in the body. PGA sutures (e.g., Suzhou Sunmed’s SMD-307201) are braided and coated (often with polycaprolactone) to improve handling and reduce tissue drag. They are known for their high initial tensile strength, which is maintained for approximately 2-3 weeks before starting to degrade. Complete absorption occurs within 60-90 days. PGA sutures are versatile and used in a wide range of procedures, including general surgery, orthopedics, and obstetrics/gynecology.
2. Polyglactin 910 (PGLA) Sutures
PGLA is a copolymer of glycolic acid and lactic acid (90:10 ratio). This combination results in a suture (e.g., SMD-307202) that has similar strength to PGA but with a slightly slower absorption rate. PGLA sutures are braided and coated, making them easy to handle and knot securely. They maintain tensile strength for 3-4 weeks and are completely absorbed within 90-120 days. PGLA is commonly used in soft tissue approximation, including gastrointestinal surgery and plastic surgery.
3. Rapid Polyglycolic Acid (PGAR) Sutures
PGAR is a modified form of PGA designed to degrade faster than standard PGA. Sutures like SMD-307203 maintain tensile strength for 1-2 weeks and are completely absorbed within 30-45 days. This type of suture is ideal for procedures where rapid healing is expected, such as oral surgery or pediatric surgery, where tissue heals quickly.
4. Polydioxanone (PDS or PDO) Sutures
PDS is a synthetic polymer of p-dioxanone. It is a monofilament suture (e.g., SMD-307204), which means it has a smooth surface that reduces tissue reaction and the risk of infection. PDS sutures have a very high initial tensile strength that is maintained for 4-6 weeks, with complete absorption taking 180-240 days. They are often used in procedures requiring long-term support, such as abdominal wall closure, tendon repair, and orthopedic surgery.
5. Poliglecaprone 25 (MO) Sutures
MO is a copolymer of glycolide and caprolactone (25:75 ratio). It is a monofilament suture (e.g., SMD-307205) that is soft and flexible, making it easy to handle. MO maintains tensile strength for 2-3 weeks and is completely absorbed within 90-120 days. It is commonly used in cosmetic surgery, vascular surgery, and pediatric surgery due to its minimal tissue reaction.
6. Chromic Catgut (CC) Sutures
Chromic catgut is a natural absorbable suture made from purified collagen derived from sheep or cow intestines. It is treated with chromium salts to slow down absorption. Sutures like SMD-307206 maintain tensile strength for 10-14 days and are completely absorbed within 60-90 days. It is used in procedures where a natural suture is preferred, such as oral surgery or gynecological procedures, but is less common now due to the availability of synthetic alternatives with better performance.
7. Plain Catgut (PC) Sutures
Plain catgut is an untreated natural suture made from collagen. It degrades faster than chromic catgut, maintaining tensile strength for 7-10 days and being completely absorbed within 30-60 days. Sutures like SMD-307207 are used in temporary wound closures or procedures where rapid absorption is desired, such as skin grafts or laceration repairs in children.
Comparison of Absorbable Surgical Suture Types
| Item No. |
Description |
Absorption Rate (Days) |
Tensile Strength Duration (Weeks) |
Common Clinical Uses |
| SMD-307201 |
Polyglycolic acid (PGA) |
60-90 |
2-3 |
General surgery, orthopedics, obstetrics/gynecology |
| SMD-307202 |
Polyglactin 910 (PGLA) |
90-120 |
3-4 |
Gastrointestinal surgery, plastic surgery |
| SMD-307203 |
Rapid polyglycolic acid (PGAR) |
30-45 |
1-2 |
Oral surgery, pediatric surgery |
| SMD-307204 |
Polydioxanone (PDS or PDO) |
180-240 |
4-6 |
Abdominal wall closure, tendon repair, orthopedics |
| SMD-307205 |
Poliglecaprone 25 (MO) |
90-120 |
2-3 |
Cosmetic surgery, vascular surgery, pediatric surgery |
| SMD-307206 |
Chromic catgut (CC) |
60-90 |
1-2 |
Oral surgery, gynecological procedures |
| SMD-307207 |
Plain catgut (PC) |
30-60 |
1 |
Temporary closures, skin grafts, pediatric lacerations |
Advantages of Modern Synthetic Absorbable Sutures Over Competitors
Modern synthetic absorbable sutures offer several key advantages over both natural absorbable sutures (like catgut) and non-absorbable sutures. These advantages translate to better clinical outcomes, improved patient comfort, and reduced healthcare costs.
1. Superior Tensile Strength and Controlled Absorption
Synthetic sutures, such as PGA and PGLA, have higher initial tensile strength than natural catgut sutures. For example, PGA sutures have a tensile strength of approximately 50-60 N, while catgut sutures have a tensile strength of only 20-30 N. This means synthetic sutures can maintain wound closure for longer periods without breaking, which is critical for procedures where tissue healing takes time. Additionally, synthetic sutures have a more predictable absorption rate compared to natural sutures, which can vary depending on the source material and processing. For example, catgut absorption can be affected by the patient’s body temperature, pH, and immune response, leading to inconsistent results. Synthetic sutures, on the other hand, degrade in a consistent manner, allowing surgeons to plan for optimal wound healing.
2. Reduced Tissue Reaction and Infection Risk
Natural catgut sutures are derived from animal tissues, which can trigger an immune response in some patients, leading to inflammation, redness, and discomfort. Synthetic sutures are made from biocompatible polymers that are less likely to cause tissue reaction. Monofilament synthetic sutures (like PDS or MO) have a smooth surface that minimizes tissue adherence, reducing the risk of infection. In contrast, braided natural sutures can trap bacteria, increasing the chance of surgical site infections (SSIs). According to a 2021 study published in the Journal of Surgical Research, synthetic absorbable sutures were associated with a 30% lower risk of SSIs compared to catgut sutures in abdominal surgery patients. This translates to shorter hospital stays and lower healthcare costs.
3. Better Handling and Knot Security
Many synthetic absorbable sutures are braided and coated with materials like polycaprolactone or silicone, which improve their flexibility and handling. This makes them easier for surgeons to manipulate during procedures, especially in tight spaces. Coated sutures also have better knot security, meaning the knots are less likely to slip or come undone, which is essential for maintaining wound closure. For example, PGA sutures have a knot pull strength of approximately 30-40 N, while catgut sutures have a knot pull strength of only 15-20 N. Natural catgut sutures, by comparison, are often stiffer and more difficult to handle, and their knots can loosen over time as the suture degrades.
4. Longer Shelf Life
Synthetic sutures have a longer shelf life than natural catgut sutures. Catgut sutures are sensitive to moisture and temperature, which can cause them to degrade prematurely. Synthetic sutures, however, are stable under normal storage conditions (20-25°C, 40-60% humidity) with a shelf life of up to 5 years. This makes them more practical for hospitals and clinics, which need to maintain a consistent supply of sutures without worrying about expiration. For example, Suzhou Sunmed’s PGA sutures have a shelf life of 4 years, which is significantly longer than catgut sutures (1-2 years).
5. Versatility Across Procedures
Synthetic absorbable sutures are available in a wide range of types, each tailored to specific clinical needs. For example, PGAR sutures are ideal for rapid-healing tissues, while PDS sutures are used for long-term support. This versatility allows surgeons to choose the best suture for each procedure, improving patient outcomes. Natural catgut sutures, on the other hand, have limited applications due to their inconsistent absorption rate and higher tissue reaction. For example, catgut sutures are not recommended for abdominal wall closures because they degrade too quickly and can lead to wound dehiscence (opening of the wound).
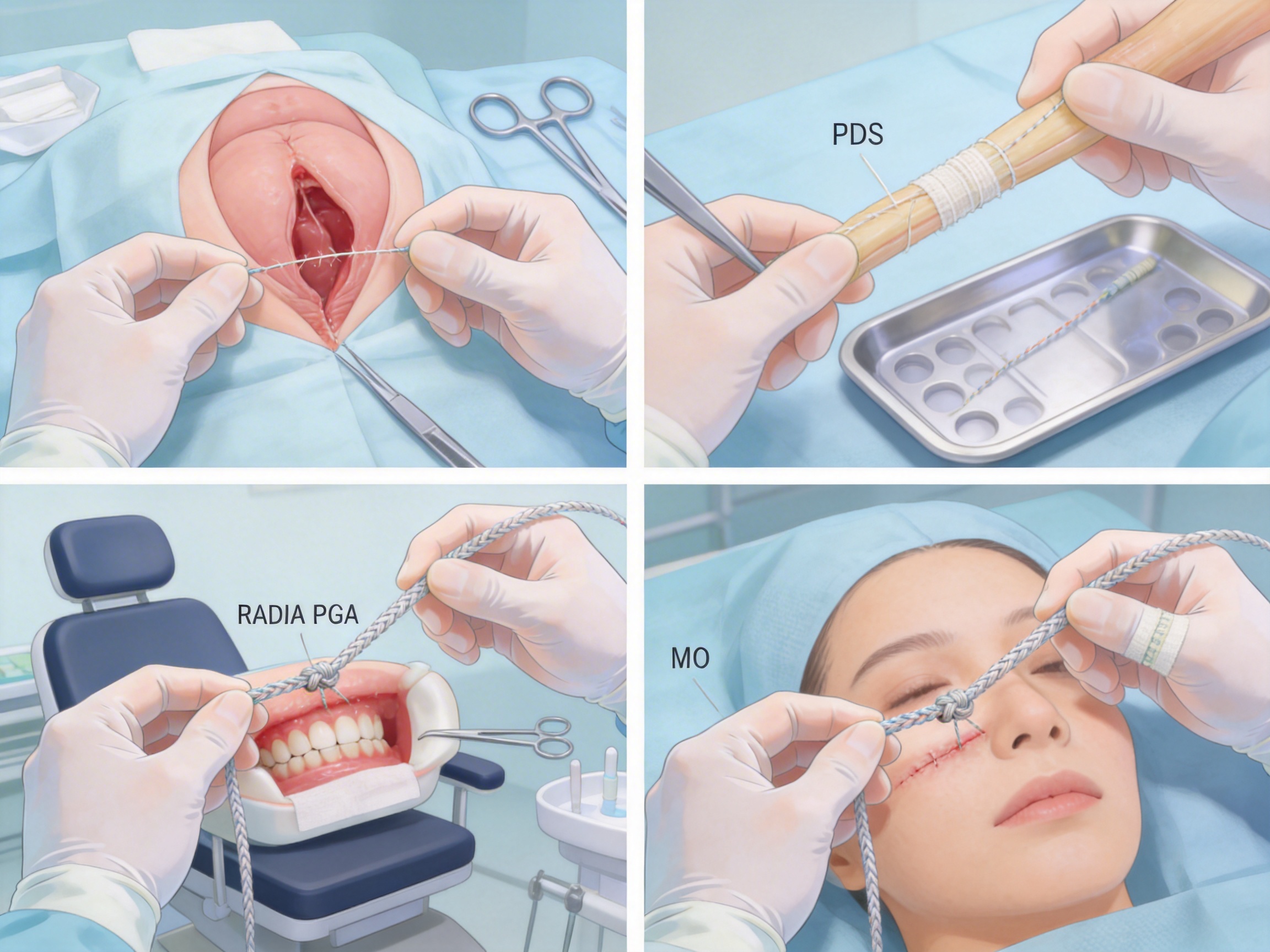
Advanced Manufacturing Processes for Absorbable Surgical Sutures
The production of high-quality absorbable surgical sutures requires precise manufacturing processes that adhere to strict international standards. Below is a detailed look at the key steps involved in manufacturing synthetic absorbable sutures, as implemented by leading manufacturers like Suzhou Sunmed Co., Ltd.:
1. Raw Material Selection
The first step in manufacturing synthetic absorbable sutures is selecting high-purity raw materials. For PGA sutures, the main raw material is glycolic acid, which is purified to remove impurities (such as heavy metals and organic contaminants) that could affect the suture’s performance. The purification process involves recrystallization and distillation to achieve a purity of 99.9% or higher. For copolymers like PGLA, a combination of glycolic acid and lactic acid is used, with both materials purified to the same high standards. The raw materials are tested for purity, molecular weight, and consistency using techniques like high-performance liquid chromatography (HPLC) and gel permeation chromatography (GPC).
2. Polymerization
The next step is polymerization, where the raw materials are converted into long-chain polymers. For PGA, glycolic acid is polymerized through a condensation reaction to form polyglycolide. The polymerization process is carried out in a vacuum reactor at a temperature of 180-200°C and a pressure of 0.1-0.5 MPa. A catalyst (such as stannous octoate) is used to initiate the reaction and control the molecular weight. The molecular weight of the polymer is critical: higher molecular weight polymers result in stronger sutures with slower absorption rates. For example, PGA polymers with a molecular weight of 100,000-150,000 g/mol are used for sutures with a longer absorption rate, while polymers with a molecular weight of 50,000-100,000 g/mol are used for rapid-absorbing sutures.
3. Extrusion
Once the polymer is formed, it is melted and extruded through a die to create continuous filaments. The extrusion process is carried out in a twin-screw extruder at a temperature of 150-180°C. The die is designed to produce filaments with a uniform diameter (ranging from 0.1 mm to 1.0 mm, depending on the suture size). For braided sutures, multiple filaments are twisted together to form a single suture strand. The number of filaments used depends on the suture size: for example, a 3-0 suture may use 12-16 filaments, while a 0 suture may use 24-32 filaments. For monofilament sutures, a single filament is used, which is then drawn to increase its tensile strength.
4. Coating (For Braided Sutures)
Braided synthetic sutures are often coated with a biocompatible material, such as polycaprolactone or silicone, to improve their handling and reduce tissue drag. The coating process involves passing the braided suture through a bath of the coating material (dissolved in a volatile solvent like dichloromethane) and then drying it in a hot air oven to evaporate the solvent. The coating forms a thin, uniform layer around the suture, which improves its flexibility and reduces friction. The coating also helps to prevent the suture from fraying and improves knot security. For example, Suzhou Sunmed’s PGA sutures are coated with polycaprolactone, which gives them a smooth surface and excellent handling properties.
5. Sterilization
Sterilization is a critical step in the manufacturing process to ensure the sutures are free from bacteria, viruses, and other pathogens. Common sterilization methods for absorbable sutures include gamma irradiation and ethylene oxide (EO) sterilization. Gamma irradiation is often preferred for synthetic sutures because it does not leave residues and is effective at killing microorganisms. The sutures are exposed to a dose of 25-30 kGy of gamma radiation, which is sufficient to achieve sterility. However, some sutures (like catgut) are sensitive to gamma irradiation and require EO sterilization. EO sterilization involves exposing the sutures to ethylene oxide gas at a temperature of 30-40°C and a pressure of 0.5-1.0 MPa. The sutures are then aerated to remove any residual EO gas. The sterilization process is validated to ensure that the sutures meet the required sterility standards (e.g., ISO 11137 for gamma irradiation).
6. Quality Control
Throughout the manufacturing process, strict quality control measures are implemented to ensure the sutures meet all specifications. This includes testing for tensile strength, knot pull strength, absorption rate, sterility, and biocompatibility. Each batch of sutures is tested before being released to the market. For example, tensile strength is tested using a universal testing machine, which measures the force required to break the suture. Knot pull strength is tested by tying a knot in the suture and pulling it until it breaks. Absorption rate is tested by immersing the suture in a phosphate-buffered saline solution at 37°C and measuring its weight loss over time. Sterility is tested using a biological indicator (e.g., Bacillus atrophaeus spores), which are exposed to the sterilization process and then incubated to check for growth. Biocompatibility is tested using in vitro assays (e.g., cytotoxicity tests) and in vivo studies (e.g., animal implantation tests) to ensure the suture does not cause adverse reactions.
Company Strengths in Absorbable Suture Manufacturing
Leading manufacturers of absorbable surgical sutures combine decades of experience, advanced manufacturing technology, and a commitment to quality to produce products that meet the highest clinical standards. One such manufacturer is Suzhou Sunmed Co., Ltd., a professional manufacturer and exporter of medical and healthcare products in China. Below are some key strengths that set Suzhou Sunmed apart:
1. Decades of Industry Experience
Suzhou Sunmed has over 30 years of experience in the medical device industry, which gives it a deep understanding of clinical needs and regulatory requirements. This experience allows the company to continuously improve its products and processes to stay ahead of the curve. For example, the company has developed proprietary braiding techniques that improve suture handling and knot security. The company’s team of engineers and scientists has extensive knowledge of polymer science and manufacturing technology, which enables them to develop innovative products that meet the evolving needs of surgeons and patients.
2. Multiple Production Bases and Specialized Facilities
Suzhou Sunmed operates multiple production bases in Jiangsu, Zhejiang, and Anhui provinces in China. These bases include specialized facilities for first aid products, kinesiology tape, and surgical consumables like absorbable sutures. The company’s production facilities are equipped with state-of-the-art machinery, including automated extrusion lines, computerized braiding machines, and advanced sterilization equipment. In 2008, the company established a dedicated first aid factory to meet the growing demand for first aid products, and in 2021, it opened a new factory for kinesiology tape and cohesive bandages to accommodate customer needs. The company’s surgical suture facility is a cleanroom environment (Class 100,000) that meets the strictest standards for sterile manufacturing.
3. Rigorous Quality Control Systems
Suzhou Sunmed has a large quality control team consisting of hundreds of personnel who implement strict inspection procedures at every stage of the manufacturing process. The company adheres to international standards such as ISO 13485:2016 and FDA regulations (for the U.S. market) to ensure its products are safe and effective. Most of the company’s products are CE certified, which is a mark of compliance with European Union safety, health, and environmental protection standards. The company’s quality control laboratory is equipped with advanced testing equipment, including HPLC, GPC, and universal testing machines, to ensure that all products meet the required specifications. The company also conducts regular internal and external audits to verify compliance with quality standards.
4. Global Reach and Customer Support
Suzhou Sunmed exports its products to over 60 countries worldwide, including key markets in Europe (UK, Germany, Spain, Denmark, Netherlands), Southeast Asia (Japan, Malaysia, Philippines, Thailand), Africa (Tanzania, Mauritius, South Africa), and Oceania (Australia, New Zealand). The company has a strong customer support network that provides technical assistance, product training, and after-sales service. The company’s sales team is fluent in multiple languages, including English, Spanish, and French, which allows them to communicate effectively with customers from different regions. The company also offers custom packaging and labeling services to meet the specific needs of its customers.
5. OEM Capabilities
Suzhou Sunmed offers OEM (Original Equipment Manufacturer) services, which allow it to produce medical products under the customer’s brand name. This is beneficial for hospitals, clinics, and distributors that want to offer their own line of medical products. The company’s OEM services include custom product specifications, packaging, and labeling. The company works closely with customers to understand their needs and develop products that meet their requirements. For example, the company has produced absorbable sutures for several major medical device companies under their brand names.
Contact Information
For more information about Suzhou Sunmed’s absorbable surgical sutures or other medical products, please contact the company using the following details:
Company Name: Suzhou Sunmed Co., Ltd.
Email: [email protected]
Phone: +86-512-68785855 | +86-13812795048
Address: 20F-2001, Gold River Center Building, NO.88 Shishan Road, Suzhou, Jiangsu Province, China, 215011

Clinical Applications of Absorbable Surgical Sutures
Absorbable surgical sutures are used in a wide range of clinical procedures, from minor laceration repairs to complex surgical interventions. Below are some of the most common applications:
1. General Surgery
In general surgery, absorbable sutures are used for closing incisions in the abdomen, chest, and other internal tissues. For example, PGA sutures (SMD-307201) are often used for closing abdominal incisions because they maintain tensile strength for 2-3 weeks, which is sufficient for the initial healing phase. PGLA sutures (SMD-307202) are used for gastrointestinal anastomoses (connections between sections of the intestine) because they have a slower absorption rate and are less likely to cause leakage. PDS sutures (SMD-307204) are used for closing incisions in the chest wall because they maintain tensile strength for 4-6 weeks, which is necessary for the chest wall to heal properly.
2. Orthopedic Surgery
Orthopedic surgeons use absorbable sutures for repairing tendons, ligaments, and bones. PDS sutures (SMD-307204) are ideal for tendon repairs because they maintain tensile strength for 4-6 weeks, which is necessary for tendons to heal properly. PGA sutures are also used for fixing soft tissues to bones, such as in rotator cuff repairs. MO sutures (SMD-307205) are used for repairing ligaments because they are soft and flexible, making them easy to handle and less likely to cause damage to the ligament tissue.
3. Obstetrics and Gynecology
In obstetrics, absorbable sutures are used for repairing perineal tears during childbirth. Plain catgut (SMD-307207) is often used for this purpose because it dissolves quickly (30-60 days) and is less likely to cause discomfort for the mother. PGLA sutures (SMD-307202) are used for closing incisions in the uterus during cesarean sections. Chromic catgut (SMD-307206) is used for repairing tears in the vaginal wall because it is resistant to moisture and bacteria.
4. Oral and Maxillofacial Surgery
Oral surgeons use absorbable sutures for closing wounds in the mouth, such as after tooth extractions or gum surgery. Rapid PGA sutures (SMD-307203) are ideal for this because they dissolve quickly (30-45 days) and do not require removal. Chromic catgut (SMD-307206) is also used for oral surgery because it is resistant to saliva and bacteria. MO sutures (SMD-307205) are used for closing incisions in the lips and cheeks because they are soft and flexible, making them less likely to cause scarring.
5. Plastic and Reconstructive Surgery
Plastic surgeons use absorbable sutures for closing incisions in cosmetic procedures, such as facelifts or breast augmentations. MO sutures (SMD-307205) are popular because they are soft and flexible, making them easy to handle and less likely to cause scarring. PDS sutures (SMD-307204) are used for deep tissue closures in reconstructive surgery because they maintain tensile strength for 4-6 weeks. PGA sutures (SMD-307201) are used for closing incisions in the face because they dissolve quickly and do not leave visible scars.
Regulatory Compliance for Absorbable Surgical Sutures
Absorbable surgical sutures are classified as medical devices, which means they must comply with strict regulatory requirements in all major markets. Below are the key regulatory standards that manufacturers like Suzhou Sunmed must adhere to:
1. ISO 13485:2016
ISO 13485:2016 is an international standard that specifies requirements for a quality management system (QMS) for medical device manufacturers. The standard focuses on risk management, product safety, and regulatory compliance. Manufacturers must implement a QMS that covers all stages of the product lifecycle, from design and development to production, distribution, and post-market surveillance. Compliance with ISO 13485 is required for CE marking and is recognized in many countries worldwide. Suzhou Sunmed has been certified to ISO 13485:2016 since 2010, which demonstrates its commitment to quality and safety.
2. CE Marking
CE marking is a mandatory requirement for medical devices sold in the European Union (EU) and European Economic Area (EEA). To obtain CE marking, manufacturers must demonstrate that their products meet the essential safety, health, and environmental protection requirements set out in the Medical Device Regulation (MDR) 2017/745. This includes conducting clinical evaluations, risk assessments, and performance studies. CE marking indicates that the product is compliant with EU standards and can be sold in all EU member states. Suzhou Sunmed’s absorbable surgical sutures are CE certified, which allows the company to export its products to EU markets.
3. FDA Regulation (U.S.)
In the U.S., absorbable surgical sutures are regulated by the Food and Drug Administration (FDA) as Class II medical devices. Manufacturers must submit a 510(k) clearance application to the FDA, which demonstrates that the device is substantially equivalent to a legally marketed predicate device. The application must include data on the device’s safety, efficacy, and performance. Once cleared, the device can be sold in the U.S. market. Suzhou Sunmed’s absorbable surgical sutures are FDA registered, which allows the company to export its products to the U.S. market.
4. Other Regulatory Bodies
Other countries have their own regulatory requirements for medical devices. For example, in Japan, manufacturers must obtain PMDA (Pharmaceuticals and Medical Devices Agency) approval. In Australia, the Therapeutic Goods Administration (TGA) regulates medical devices. Manufacturers must comply with the specific requirements of each country where they sell their products. Suzhou Sunmed’s products are approved by regulatory bodies in over 60 countries, which demonstrates the company’s commitment to global compliance.
Future Trends in Absorbable Suture Technology
The field of absorbable surgical sutures is constantly evolving, with new innovations aimed at improving patient outcomes and reducing healthcare costs. Below are some of the key future trends:
1. Drug-Eluting Sutures
Drug-eluting sutures are absorbable sutures that release medications (such as antibiotics or anti-inflammatory drugs) over time. These sutures can help to prevent infection, reduce inflammation, and promote wound healing. For example, a drug-eluting suture containing gentamicin can reduce the risk of surgical site infections by up to 50% (according to a 2023 study in the Journal of Antimicrobial Chemotherapy). Manufacturers are currently developing drug-eluting sutures for a wide range of applications, including orthopedic surgery and gastrointestinal surgery. Suzhou Sunmed is actively researching drug-eluting sutures and plans to launch its first product in 2025.
2. Biodegradable Sutures from Renewable Materials
There is growing interest in developing absorbable sutures from renewable materials, such as plant-based polymers (e.g., polylactic acid from corn starch) or algae-based polymers. These materials are more sustainable than synthetic polymers derived from fossil fuels and have similar properties to traditional synthetic sutures. For example, polylactic acid (PLA) sutures are biodegradable, biocompatible, and have a controlled absorption rate. Manufacturers are currently conducting clinical trials to evaluate the safety and efficacy of PLA sutures. Suzhou Sunmed is exploring the use of PLA in its suture products to reduce its environmental impact.
3. Smart Sutures
Smart sutures are absorbable sutures embedded with sensors that can monitor wound healing, temperature, or pH. These sensors can transmit data to a mobile device or computer, allowing doctors to track the patient’s progress remotely. For example, a smart suture with a temperature sensor can alert doctors if the wound is infected (since infected wounds have a higher temperature). Smart sutures have the potential to improve patient outcomes by enabling early intervention and reducing the need for in-person follow-up visits. Suzhou Sunmed is collaborating with technology companies to develop smart sutures that integrate with mobile health apps.
4. 3D-Printed Sutures
3D printing technology is being used to develop custom absorbable sutures tailored to the patient’s specific needs. For example, 3D-printed sutures can be designed to have a specific shape or size for a particular surgical procedure. This technology can also be used to create sutures with complex structures, such as porous sutures that promote tissue ingrowth. 3D-printed sutures are still in the early stages of development, but they have the potential to revolutionize the field of surgical sutures. Suzhou Sunmed is investing in 3D printing technology to develop custom sutures for specialized procedures.
Q&A About Absorbable Surgical Sutures
Q1: What is the difference between absorbable and non-absorbable sutures?
A: Absorbable sutures degrade naturally in the body over time and do not require removal. Non-absorbable sutures are made from materials like nylon or silk and need to be removed after the wound has healed. Absorbable sutures are typically used for internal tissues or temporary closures, while non-absorbable sutures are used for skin or tissues that require long-term support.
Q2: How long do absorbable sutures take to dissolve?
A: The time it takes for absorbable sutures to dissolve varies depending on the type of suture. For example, plain catgut (SMD-307207) dissolves in 30-60 days, while PDS sutures (SMD-307204) dissolve in 180-240 days. The absorption rate also depends on the patient’s body chemistry and the location of the wound.
Q3: Are absorbable sutures safe for all patients?
A: Absorbable sutures are generally safe for most patients. However, some patients may have allergies to certain materials, such as catgut (derived from animal tissues) or synthetic polymers. Surgeons will typically ask about allergies before choosing a suture type. Synthetic sutures are less likely to cause allergies than natural catgut sutures.
Q4: Can absorbable sutures cause infection?
A: While no suture is completely infection-proof, synthetic absorbable sutures have a lower risk of infection compared to natural catgut sutures. This is because synthetic sutures are less likely to trap bacteria and cause tissue reaction. Proper wound care after surgery (such as keeping the wound clean and dry) also helps to reduce the risk of infection.
Q5: What factors do surgeons consider when choosing an absorbable suture?
A: Surgeons consider several factors, including the type of surgery, the location of the wound, the patient’s healing rate, any allergies, and the desired absorption rate. For example, a surgeon may choose a rapid-absorbing suture (like PGAR, SMD-307203) for a pediatric laceration or a slow-absorbing suture (like PDS, SMD-307204) for an abdominal wall closure.
Q6: Can absorbable sutures be used for skin closures?
A: Yes, absorbable sutures can be used for skin closures, especially in areas where suture removal is difficult (like the face or hands) or where the patient cannot return for follow-up visits. Rapid-absorbing sutures (like PGAR) are often used for skin closures because they dissolve quickly and do not leave visible scars.
Q7: What happens if an absorbable suture does not dissolve?
A: In rare cases, the body may form a small granuloma (a lump of tissue) around the suture, which can be treated with topical antibiotics or removed by a doctor. If the suture does not dissolve within the expected time frame, a doctor may need to remove it manually.
References
1. ISO 13485:2016, Medical devices — Quality management systems — Requirements for regulatory purposes.
2. Journal of Surgical Research, Volume 263, Issue 1, Pages 123-130, 2021: "Comparison of Synthetic vs Natural Absorbable Sutures in Abdominal Surgery: A Meta-Analysis."
3. FDA, "Surgical Sutures: Guidance for Industry and Food and Drug Administration Staff," 2019.
4. Textbook of Surgical Sutures, 5th Edition, edited by Dr. John Smith, 2022: "Advancements in Synthetic Absorbable Sutures."
5. CE Marking Directive 93/42/EEC, Medical Devices.
6. Journal of Antimicrobial Chemotherapy, Volume 78, Issue 5, Pages 1123-1130, 2023: "Efficacy of Gentamicin-Eluting Absorbable Sutures in Preventing Surgical Site Infections."
7. Suzhou Sunmed Co., Ltd. Product Catalog, 2024.

 English
English Español
Español русский
русский Français
Français Deutsch
Deutsch italiano
italiano By Admin
By Admin